Hysterectomy has been done vaginally since 120 A.D., and through a laparotomy, as with a c-section, since the mid 1800s, centuries before the advent of the morcellator. 92% of hysterectomies are elective surgeries for benign conditions: fibroids, heavy bleeding, endometriosis. Though these conditions are painful without question, none represent conditions we'd risk upstaging cancer to an incurable level for - agreed?
As far as the way the surgery is performed goes, there are no circumstances under which a morcellator, a stick blender-like device used to chop up the uterus so it fits out the holes of a laparoscopic surgery, is required. More women agree to surgery when it is referred to as "minimally-invasive," sure, but the question is whether they would agree to this if they knew it increased their risk of death.
An even better question is why doctors would propose it, given how obvious it is that grinding up potential cancer inside the body can never be preferable to removing it intact. Yet, that's what's been done for a couple of decades now, and that's what continues, even after the well-covered case of Dr. Amy Reed, brought to light through her tireless advocacy and that of her husband, Dr. Hooman Noorchashm, as she fights her upstaged cancer and they continue to work and to raise their 6 children.
Because they pressed the issue, the FDA reworked the ACOG's math. The ACOG had been saying that the risk of a fibroid turning out to be cancer was 1 in 10,000. In fact, the FDA concluded, the risk is 1 in 352. The ACOG hadn't been using the right denominator to arrive at the risk. They'd been using "all women in the U.S." versus "all women who presented for surgery with fibroids."
Even according to the ACOG's own guidelines for when to perform a hysterectomy, this surgery is performed far too often. Other types of surgeons, those who are trained in general surgery (OBGYNs are not), remove illness, not entire organs. In the cases where women's fibroids turned out instead to have been sarcoma (cancer), had the cancer been removed in place ("in situ"), it certainly would not have been upstaged in the manner in which it is when a morcellator is used.
As you're reading these notes, which are directly quoted from the ACOG's May 2015 Annual Meeting, it's helpful to be aware that Dr. Keith Isaacson was a member of the panel that heard women's testimony to the FDA in 2014 about the fact that the women's cancer had been rendered incurable through the use of morcellation. Dr. Isaacson was allowed to remain on the panel despite the fact that he had a known financial conflict, having received money from robotic surgery device makers. You can read about that here.
The original ACOG meeting notes can be found here.
Directly quoted from the ACOG's May 2015 Annual meeting:
"Mathers Lecture to Focus on Power Morcellation Concerns Posted By: ACOG on: May 03, 2015 This year’s John and Marney Mathers Lecture will consider the risks and rewards of power morcellation.
“Morcellation Debate,” coming on the heels of last year’s FDA safety communication about the procedure, takes place at 3:15 pm today in Hall D and features Keith B. Isaacson, director of minimally invasive gynecologic surgery and infertility and Newton-Wellesley Hospital in Boston, and Javier F. Magrina, MD, professor of obstetrics-gynecology at the Mayo Clinic in Arizona. They will debate power morcellation’s benefits, concerns, sequelae, prevention of complications and the use of alternate techniques.
The communication, first released in April 2014 and updated in November, warned against using laparoscopic power morcellators in most patients undergoing hysterectomy or myomectomy for uterine fibroids because of a risk of spreading unsuspected cancerous tissue, notably uterine sarcomas, outside the uterus. Contraindications include suspicion of cancer, menopause and small uterus.
The risk of sarcoma in a fibroid exists, and morcellation of that fibroid may spread and upstage the disease, Dr. Isaacson said, but the treatment decision should rest with the patient and her health-care provider.
“We just don’t have enough additional information to say that this is considered bad medicine because there are many advantages to a smaller incision, depending upon the patient’s preferences and her lifestyle and priorities, she may elect to do that,” said Dr. Isaacson, who was on the FDA panel that looked into power morcellation but didn’t work on the final document.
The FDA document lists several contraindications. But for many younger or perimenopausal women, the procedure has great benefits that must be considered.
The FDA and physician statements from ACOG, the AAGL and other organizations that have examined existing data still provide an opportunity for the informed patient to make the final call.
“I don’t care what number you pick. Let’s say the risk of cancer is 1 in 10 instead of 1 in 8,000,” he said. “I still think the patient should be involved in the decision-making process after a discussion of the benefits vs. the risks.”
Dr. Magrina opposes any morcellation that takes place outside an endobag. He will argue that morcellation safety will improve with use of endobags to contain the morcellated material.
“Stopping only power morcellation doesn’t stop the problems that happen with morcellation by other means, such as using a regular cold knife if what you’re morcellating is exposed to the intraperitoneal cavity,” he said.
Dr. Magrina said that the FDA missed an opportunity in its safety communication by not addressing the use of endobags. More companies are working with contained morcellation concepts that will improve patient safety.
“I’m actually con and pro depending on how you do it — contained or uncontained,” he said. “The point that I’m making has never been discussed at the FDA level.”
While concerns about spreading and upstaging cancer have grabbed most of the attention, Dr. Magrina said that more people develop benign conditions such as endometriosis and peritoneal leiomyomatosis that spread into the peritoneal cavity during morcellation done without an endobag.
“The problem is not only with cancer,” he said. “And it makes no difference whether the morcellation is by knife or power morcellator.”
As far as the way the surgery is performed goes, there are no circumstances under which a morcellator, a stick blender-like device used to chop up the uterus so it fits out the holes of a laparoscopic surgery, is required. More women agree to surgery when it is referred to as "minimally-invasive," sure, but the question is whether they would agree to this if they knew it increased their risk of death.
An even better question is why doctors would propose it, given how obvious it is that grinding up potential cancer inside the body can never be preferable to removing it intact. Yet, that's what's been done for a couple of decades now, and that's what continues, even after the well-covered case of Dr. Amy Reed, brought to light through her tireless advocacy and that of her husband, Dr. Hooman Noorchashm, as she fights her upstaged cancer and they continue to work and to raise their 6 children.
Because they pressed the issue, the FDA reworked the ACOG's math. The ACOG had been saying that the risk of a fibroid turning out to be cancer was 1 in 10,000. In fact, the FDA concluded, the risk is 1 in 352. The ACOG hadn't been using the right denominator to arrive at the risk. They'd been using "all women in the U.S." versus "all women who presented for surgery with fibroids."
Even according to the ACOG's own guidelines for when to perform a hysterectomy, this surgery is performed far too often. Other types of surgeons, those who are trained in general surgery (OBGYNs are not), remove illness, not entire organs. In the cases where women's fibroids turned out instead to have been sarcoma (cancer), had the cancer been removed in place ("in situ"), it certainly would not have been upstaged in the manner in which it is when a morcellator is used.
As you're reading these notes, which are directly quoted from the ACOG's May 2015 Annual Meeting, it's helpful to be aware that Dr. Keith Isaacson was a member of the panel that heard women's testimony to the FDA in 2014 about the fact that the women's cancer had been rendered incurable through the use of morcellation. Dr. Isaacson was allowed to remain on the panel despite the fact that he had a known financial conflict, having received money from robotic surgery device makers. You can read about that here.
The original ACOG meeting notes can be found here.
Directly quoted from the ACOG's May 2015 Annual meeting:
"Mathers Lecture to Focus on Power Morcellation Concerns Posted By: ACOG on: May 03, 2015 This year’s John and Marney Mathers Lecture will consider the risks and rewards of power morcellation.
“Morcellation Debate,” coming on the heels of last year’s FDA safety communication about the procedure, takes place at 3:15 pm today in Hall D and features Keith B. Isaacson, director of minimally invasive gynecologic surgery and infertility and Newton-Wellesley Hospital in Boston, and Javier F. Magrina, MD, professor of obstetrics-gynecology at the Mayo Clinic in Arizona. They will debate power morcellation’s benefits, concerns, sequelae, prevention of complications and the use of alternate techniques.
The communication, first released in April 2014 and updated in November, warned against using laparoscopic power morcellators in most patients undergoing hysterectomy or myomectomy for uterine fibroids because of a risk of spreading unsuspected cancerous tissue, notably uterine sarcomas, outside the uterus. Contraindications include suspicion of cancer, menopause and small uterus.
The risk of sarcoma in a fibroid exists, and morcellation of that fibroid may spread and upstage the disease, Dr. Isaacson said, but the treatment decision should rest with the patient and her health-care provider.
“We just don’t have enough additional information to say that this is considered bad medicine because there are many advantages to a smaller incision, depending upon the patient’s preferences and her lifestyle and priorities, she may elect to do that,” said Dr. Isaacson, who was on the FDA panel that looked into power morcellation but didn’t work on the final document.
The FDA document lists several contraindications. But for many younger or perimenopausal women, the procedure has great benefits that must be considered.
The FDA and physician statements from ACOG, the AAGL and other organizations that have examined existing data still provide an opportunity for the informed patient to make the final call.
“I don’t care what number you pick. Let’s say the risk of cancer is 1 in 10 instead of 1 in 8,000,” he said. “I still think the patient should be involved in the decision-making process after a discussion of the benefits vs. the risks.”
Dr. Magrina opposes any morcellation that takes place outside an endobag. He will argue that morcellation safety will improve with use of endobags to contain the morcellated material.
“Stopping only power morcellation doesn’t stop the problems that happen with morcellation by other means, such as using a regular cold knife if what you’re morcellating is exposed to the intraperitoneal cavity,” he said.
Dr. Magrina said that the FDA missed an opportunity in its safety communication by not addressing the use of endobags. More companies are working with contained morcellation concepts that will improve patient safety.
“I’m actually con and pro depending on how you do it — contained or uncontained,” he said. “The point that I’m making has never been discussed at the FDA level.”
While concerns about spreading and upstaging cancer have grabbed most of the attention, Dr. Magrina said that more people develop benign conditions such as endometriosis and peritoneal leiomyomatosis that spread into the peritoneal cavity during morcellation done without an endobag.
“The problem is not only with cancer,” he said. “And it makes no difference whether the morcellation is by knife or power morcellator.”
In this 2014 video, you can hear the doctors explain that they would all perform morcellation. "The current situation with morcellation has not changed my practice in any way."
So, what are these OBGYNs saying about endobags to contain the pieces and what are they saying about screening beforehand?
Dr. Reed was carefully screened before her surgery.
Any use of an endobag is an experiment at the patient's own risk, designed only to allow OBGYNs to keep using unsafe devices.
According to the ACOG's own May 2014 Morcellation special report, endobags can tear or block the surgeons view: "Use of a Bag During Morcellation in Gynecologic SurgerySome investigators have suggested that the use of an intra-peritoneal bag during manual or power morcellation may be helpful in reducing intraperitoneal tissue dissemination (8, 30). However, power morcellation performed within a bag is not well studied and has several limitations that potentially increase the risk of the procedure. For example, currently available bags were not designed specifically for use in conjunction with power morcellation. The bags often have size limitations and have not been adequately constructed to prevent tearing by the morcellator. Further, the use of bags limits simultaneous visualization of the tissue being morcellated and the surrounding tissue that must be protected from the sharp morcellator blade."
The third doctor in the video says that if a patient is at "low risk" of cancer, he would "absolutely" use a morcellator. Is that good enough for you? In Boston, hospitals disagree as to policy.
Now imagine if, instead of figuring out better ways of dealing with uterine sarcoma, surgeons were sitting around constructing studies about how much it would cost not in lives, but in money, not to keep doing it? At Northwestern, that's exactly what they did. Is that how you imagine residents and surgeons spending their time?
"Conclusions:
Eliminating morcellation hysterectomy as a treatment for myomas is not cost-effective under a wide variety of probability and cost assumptions. Performing laparotomy for all patients who might otherwise be candidates for morcellation hysterectomy is a costly policy from a societal perspective."
This isn't the first study of its kind, either. The device manufacturer, Ethicon, sponsored this one in 2011 before the doctors broke their story.
Some OBGYN device advocates think it's ok to argue that we can't let go of technology. Though hospitals have invested heavily in these robots, that's not an excuse to keep using them if they're unsafe.
In terms of policy, who makes policy if not the FDA? Their conclusion was to put a warning on the device, but they chose not to ban it.
What about the ACOG? The ACOG's advice to its 58,000 OBGYN members was to continue to use the device, but, as usual, to put the onus back on patients. Their Committee Opinion in May 2014 was to inform patients of risk and to create a national registry of women made ill by the device. Does that make sense to you, as a patient?
Once a physician suggests performing a surgery a certain way, doesn't that bias a patient even if only by making the patient consider that option? Would a patient imagine that something being offered could take something bad and make it worse?
"Conclusions:
Eliminating morcellation hysterectomy as a treatment for myomas is not cost-effective under a wide variety of probability and cost assumptions. Performing laparotomy for all patients who might otherwise be candidates for morcellation hysterectomy is a costly policy from a societal perspective."
This isn't the first study of its kind, either. The device manufacturer, Ethicon, sponsored this one in 2011 before the doctors broke their story.
Some OBGYN device advocates think it's ok to argue that we can't let go of technology. Though hospitals have invested heavily in these robots, that's not an excuse to keep using them if they're unsafe.
In terms of policy, who makes policy if not the FDA? Their conclusion was to put a warning on the device, but they chose not to ban it.
What about the ACOG? The ACOG's advice to its 58,000 OBGYN members was to continue to use the device, but, as usual, to put the onus back on patients. Their Committee Opinion in May 2014 was to inform patients of risk and to create a national registry of women made ill by the device. Does that make sense to you, as a patient?
Once a physician suggests performing a surgery a certain way, doesn't that bias a patient even if only by making the patient consider that option? Would a patient imagine that something being offered could take something bad and make it worse?
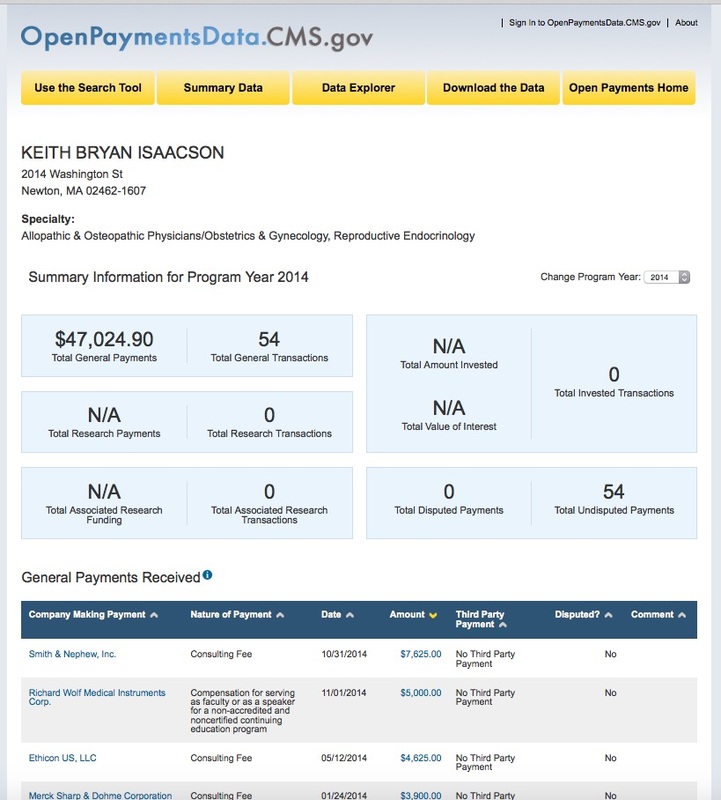
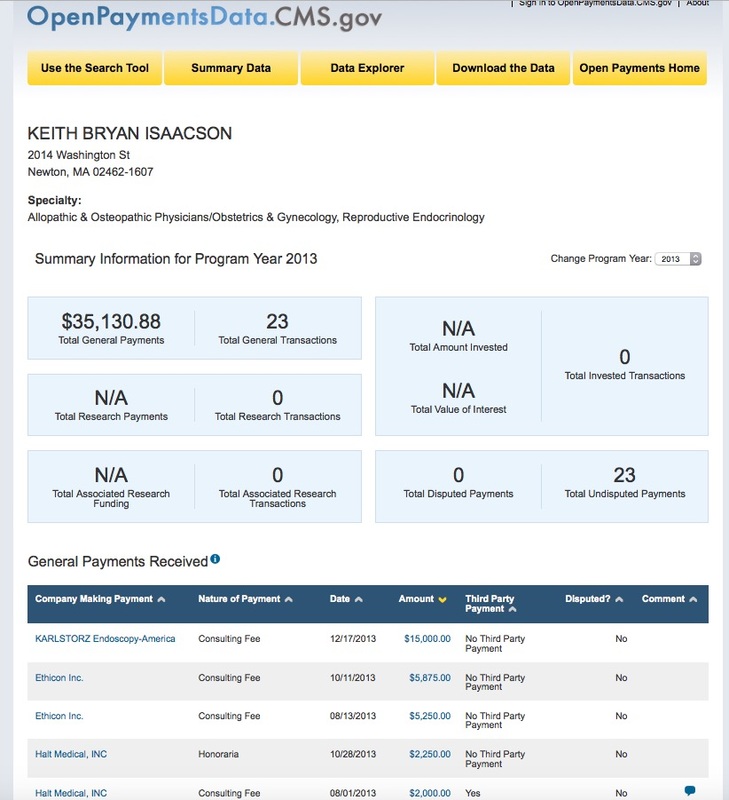
So, again, Dr. Keith Isaacson from Newton-Wellesley Hospital, who "doesn't care if 1 in 10 fibroids is sarcoma," is in favor of morcellation.
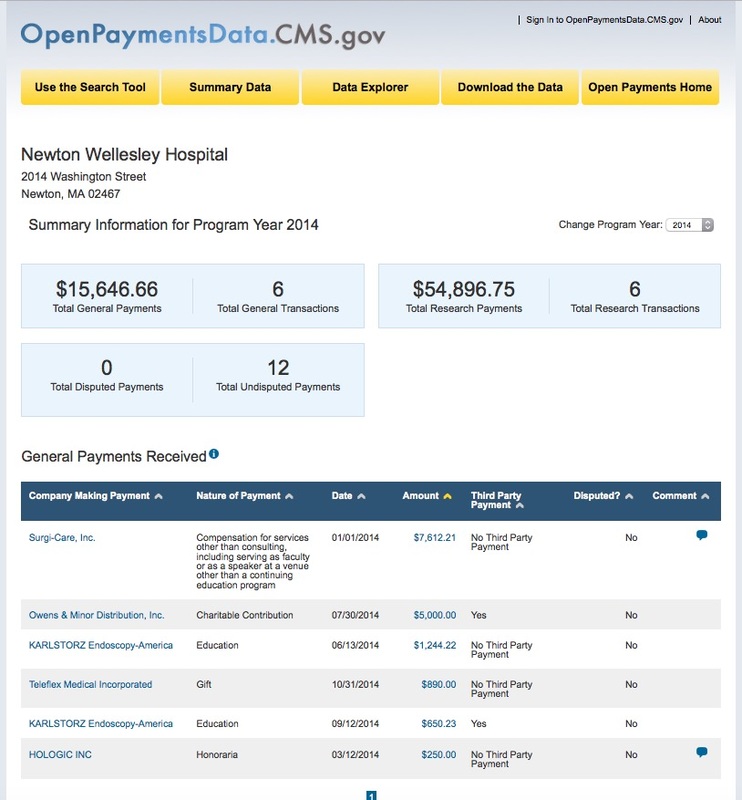
As with study results, it's important to follow the money:
As with study results, it's important to follow the money:
Dr. Javier Magrina of the Mayo Clinic was less enthusiastic. He'll do the surgery, but only using an endobag. He was also paid less by morcellation manufacturers.
Read more at Drs. Reed and Noorchashm's page, here.


 RSS Feed
RSS Feed
